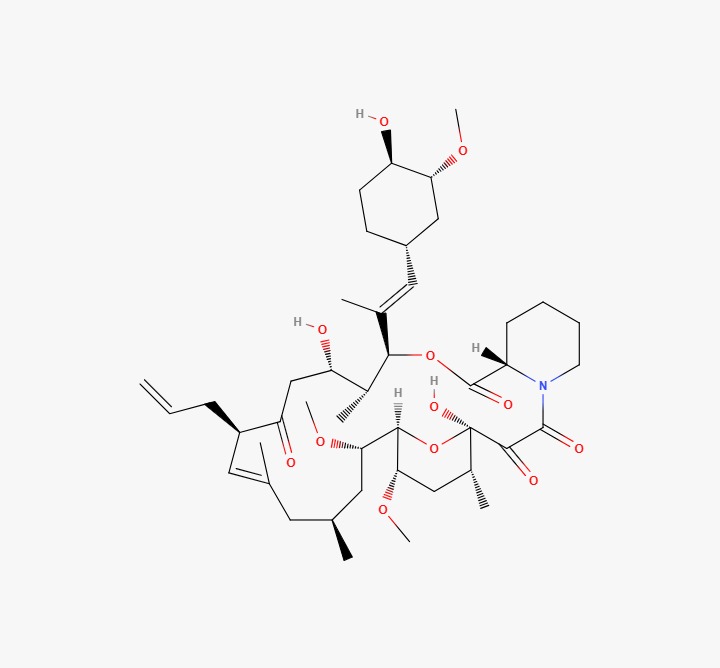
Tacrolimus API Product Category: Active Pharmaceutical Ingredient (API) CAS Number: 104987-11-3 Molecular Formula: C44H69NO12 Molecular Weight: 804.02 g/mol Grade: Pharmaceutical Grade (USP / BP / Ph.Eur / IP – as applicable) Product Overview Tacrolimus is a macrolide immunosuppressant widely used in the formulation of oral, injectable, and topical dosage forms. It is primarily indicated for the prevention of organ transplant rejection and for certain approved dermatological and autoimmune conditions. Tacrolimus API is utilized by pharmaceutical manufacturers serving regulated and semi-regulated markets globally. Applications Tacrolimus is indicated for: Prevention of organ transplant rejection Management of moderate to severe atopic dermatitis (topical formulations) Other approved immunosuppressive therapeutic indications All applications must comply with approved labeling and national regulatory guidelines. Quality & Compliance Our Tacrolimus API is sourced from GMP-compliant manufacturing facilities with robust quality assurance systems and validated production processes. Each batch is supplied with: Certificate of Analysis (COA) Material Safety Data Sheet (MSDS / SDS) Batch traceability documentation Stability data (upon request) Pharmacopoeial compliance documentation DMF support (where available) Packaging & Supply Export-grade, moisture-controlled and light-protected packaging HDPE or fiber drums with double inner liners Customized labeling and complete export documentation Bulk supply for domestic and international markets Regulatory Notice Tacrolimus is a prescription pharmaceutical ingredient. Buyers are responsible for ensuring compliance with applicable licensing requirements, regulatory approvals, and import regulations in their respective jurisdictions.