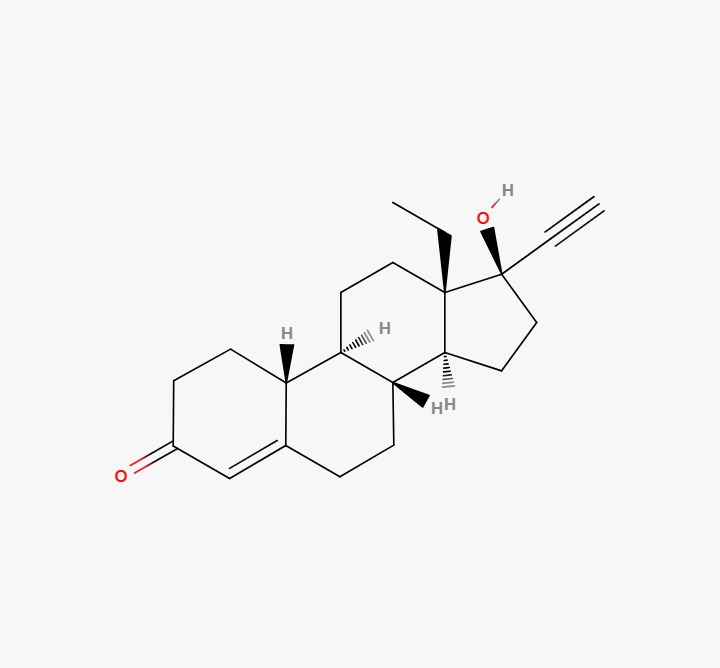
Levonorgestrel API Product Category: Active Pharmaceutical Ingredient (API) CAS Number: 797-63-7 Molecular Formula: C21H28O2 Molecular Weight: 312.45 g/mol Grade: Pharmaceutical Grade (USP / BP / Ph.Eur / IP – as applicable) Product Overview Levonorgestrel is a synthetic progestogen widely used in the formulation of oral contraceptive tablets, emergency contraceptive products, and long-acting hormonal delivery systems. It is recognized for its established therapeutic profile and stability in solid dosage formulations. Levonorgestrel API is supplied to pharmaceutical manufacturers serving regulated and semi-regulated markets under strict quality and compliance standards. Applications Levonorgestrel is indicated for: Oral contraceptive formulations Emergency contraception Hormone-based therapeutic applications Other approved gynecological indications as permitted by regulatory authorities All applications must comply with approved labeling and national regulatory guidelines. Quality & Compliance Our Levonorgestrel API is sourced from GMP-compliant manufacturing facilities with validated quality assurance systems. Each batch is supplied with: Certificate of Analysis (COA) Material Safety Data Sheet (MSDS / SDS) Batch traceability documentation Stability data (upon request) Pharmacopoeial compliance documentation DMF support (where available) Packaging & Supply Export-grade, moisture-controlled packaging HDPE or fiber drums with double inner liners Customized labeling and complete export documentation Bulk supply for domestic and international markets Regulatory Notice Levonorgestrel is a prescription pharmaceutical ingredient subject to regulatory controls in many jurisdictions. Buyers are responsible for ensuring compliance with applicable licensing requirements, regulatory approvals, and import regulations prior to procurement.